Directing neural stem cells using a microfluidic device

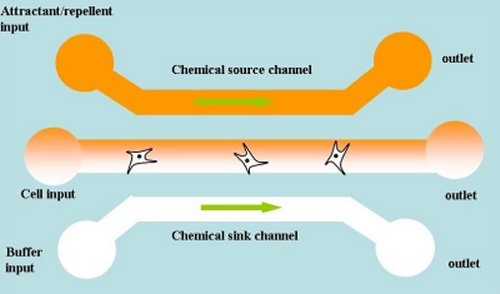
Engraftment of neural stem cells (NSCs) to replace central nervous system cells lost to injury or disease is a promising treatment strategy. However, many regions of the adult brain shown low levels of permissiveness to NSC migration, limiting the effectiveness of both endogenous and transplanted NSC therapies.
By enhancing the motility of the ‘stem cell pool’ to the site of central nervous system(CNS) injury or disease using chemical cues, treatment strategies will become more successful. Epidermal growth factor receptor (EGFR) signaling is involved in the activation of a number of downstream pathways that enhance the motility of NSCs, and therefore may represent an effective target for regenerative therapy in transplantation paradigms. There's a lot that's not known about how NSCs can be activated to replace damaged tissue in the CNS, however.
As a start, we have developed a micro-chemotaxis device that is able to provide a linear chemical concentration gradient in a microchannel with no through flow. Our device is also compatible with 3D scaffolding gels, enabling in vivo-like imaging of EGFR-enhanced NSC migration and invasiveness.
Publications:
- Shing-Yi Cheng, Steve H. Heilman, Peng Zhou, Shivaun Archer, Michael L. Shuler and Mingming Wu. A Hydrogel-based Microfluidic Device for the Studies of Bacterial and Mammalian Chemotaxis. Lab on a Chip, [submitted]
- John A. Boockvar, Dmitri Kapitonov, Gurpreet Kapoor, Joost Schoutena, George J. Counelisa, Oliver Boglerc, Evan Y. Snyderd, Tracy K. McIntosha and Donald M. O'Rourke. Constitutive EGFR Signaling Confers a Motile Phenotype to Neural Stem Cells. Mol Cell Neurosci., 2003 Dec;24(4):1116-30.
Site created and maintained by Young Joon Suh

