Interstitial Flows on Cancer Cell Invasion Revealed by 3D Microfluidics
The goal of this research project is to investigate whether interstitial flows promote cancer cell invasion. Interstitial flows are particularly important for tumor cell invasion because it is elevated in tumor microenvironment due to the heightened interstitial fluid pressure as well as the abnormal angiogenic/lymphanogenic blood/lymphatic vessels. The flow speed associated with interstitial flows are in the order of a few micrometers per second in normal tissue.
Here, we use a robust 3D microfluidic device to study the roles of interstitial fluid flows in the migration of human breast tumor cells (see set up in Figure A). We demonstrated that fluid flows at a speed of 2.0 micrometer per second promote amoeboid cell morphology and motility. Cells with aspect ratio greater than 2.0 are considered elongated or mesenchymal and cells with aspect ratio smaller than 2.0 are considered rounded or amoeboid (see definition in Figure B). We observed that the percentage of elongated cells is 58% in the absence of flows, in contrast to 27% of elongated cells in the presence of flows (see Figure C). This result is consistent with previous in vivo observation in which invasive cancer cells often exhibit amoeboid mode of migration.
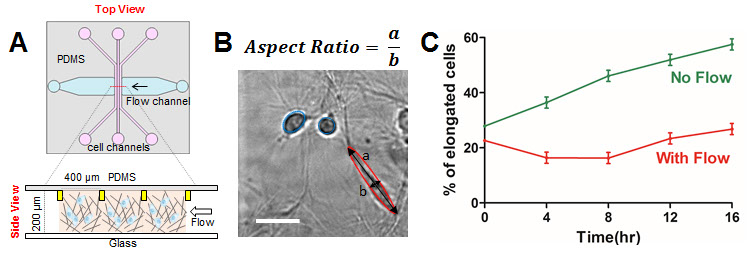
A.One of the microfluidics device with cell embedded collagen seeded in the three parallel cell channels, and fluid flows are introduced in the horizontal flow channel. Collagens are confined within the three cell channels using the contact lines, indicated by yellow rectangles.B. Micrograph of MDA-MB-231 cells. The aspect ratio of each cell is defined as the ratio of the long axis a over the short axis b. Scale bar is 100 micrometer. C. Percentage of elongated cells versus time in the absence and presence of flow.
Publications:
- Chih-kuan Tung, Oleh Krupa, Elif Apyadin, Jr-Jiun Liou, Anthony Diaz-Santana, Beum Jun Kim, and Mingming Wu, A contact line pinning based microfluidic platform for modeling physiological flows, Lab-on-a-chip, 13(19):3876-3885 (2013).
Site created and maintained by Young Joon Suh

